Yuchin Albert Pan, Ph.D.

“Because people and fish share similarities in brain structure and genes, this simple organism provides us with an extraordinary window to understand the common neural pathway and brain activity patterns.”
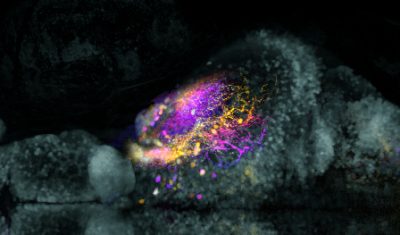
Visualizing neural networks to understand disease
What can the zebrafish brain teach us about human brain development?
The Pan lab uses zebrafish as the tool to discover the general principles of vertebrate nervous system development. These general principles will help us understand and identify novel treatment options for human neuropsychiatric disorders.
During development, neurons establish connections with each other called synapses to form functional neural circuits. Assembly of these circuits is essential for normal behavior and neuropsychiatric diseases. But the immense structural complexity of the human brain – with 80 billion neurons and trillions of synapses – makes studying developmental mechanisms and connectivity a challenge. The optically translucent Zebrafish brain, with about 100,000 neurons in the larvae, is a useful model. More than 70 percent of human disease genes have functional homologs in zebrafish, making it possible to model human mutations in this more accessible system
Using zebrafish, the Pan lab has (1) identified potential causes of saccade failure, characterized by the loss of fast eye movements (Ma et al. Journal of Neuroscience 2020), (2) uncovered the molecular mechanisms for craniofacial deficits in undiagnosed human disease (Ates et al. Disease Models and Mechanisms 2020), and (3) developed new viral vector-based tools for visualizing novel neural connectivity patterns (Ma, Kler, et al. Frontiers in Neural Circuits 2020).
The current focus of the lab is on how intercellular signaling affects the organism’s response to stressors, and how genetic deficiencies may contribute to diseases such as epilepsy and stress-associated disorders such as anxiety and depression.
- Associate Professor and Commonwealth Research Commercialization Fund Eminent Research Scholar in Developmental Neuroscience
- Associate Professor, Department of Biomedical Sciences and Pathobiology, Virginia-Maryland College of Veterinary Medicine
- Associate Professor, Department of Psychiatry and Behavioral Medicine, School of Medicine
Ma M, Ramirez AD, Wang T, Roberts RL, Harmon KE, Schoppik D, Sharma A, Kuang C, Goei SL, Gagnon JA, Zimmerman S, Tsai SQ, Reyon D, Joung JK, Aksay ERF, Schier AF, Pan YA. Zebrafish dscaml1 Deficiency Impairs Retinal Patterning and Oculomotor Function. J Neurosci. 2020;40(1):143-58. Epub 2019/11/07. doi: 10.1523/JNEUROSCI.1783-19.2019. PMCID: PMC6939486.
Ates KM, Wang T, Moreland T, Veeranan-Karmegam R, Ma M, Jeter C, Anand P, Wenzel W, Kim HG, Wolfe LA, Stephen J, Adams DR, Markello T, Tifft CJ, Settlage R, Gahl WA, Gonsalvez GB, Malicdan MC, Flanagan-Steet H, Pan YA. Deficiency in the endocytic adaptor proteins PHETA1/2 impairs renal and craniofacial development. Disease Models & Mechanisms. 2020;13(5). Epub 2020/03/11. doi: 10.1242/dmm.041913.
Ma M, Kler S, Pan YA. Structural Neural Connectivity Analysis in Zebrafish With Restricted Anterograde Transneuronal Viral Labeling and Quantitative Brain Mapping. Frontiers in Neural Circuits. 2019;13(85):85. Epub 2020/02/11. doi: 10.3389/fncir.2019.00085. PMCID: PMC6989443.
- Medical College of Georgia, Augusta University
Assistant Professor, Department of Neuroscience and Regenerative Medicine, Department of Neurology
Co-Director, Department of Neuroscience and Regenerative Medicine Microscopy Facility
Co-Director, Transgenic Zebrafish Core Laboratory
Affiliate, James & Jean Culver Vision Discovery Institute - Harvard University
Postdoctoral Fellow, Department of Molecular and Cellular Biology
Postdoctoral Fellow, Center for Brain Science
- Washington University School of Medicine, St. Louis: Ph.D., Neural Development
- National Taiwan University: B.S., Zoology
- First Place, Marine Biological Laboratory Photomicrography Contest, 2010
- Image of Distinction, Nikon Small World Photomicrography Competition, 2009
- Fourth place, Olympus Bioscapes Digital Imaging Competition, 2008
- Ruth L. Kirschstein National Research Service Awards for Individual Postdoctoral Fellow, 2007
- Finalist, Upstate Young Cell Signaler Award, 2005
- Ruth L. Kirschstein National Research Service Awards for Individual Predoctoral Fellow, 2004
- Honorable Mention, National Science Foundation Graduate Fellowship, 2001
-
Article Item
-
Article Item
-
Article ItemFralin Biomedical Research Institute at VTC scientists identify beacon molecule that prevents vision, behavioral problems in mice , article
The findings, published in the Proceedings of the National Academy of Science, help decode how these complicated brain circuits form, laying a potential foundation for future research into how these pathways may be coaxed to regenerate.
Date: Oct 20, 2021 -
Article ItemFralin Biomedical Research Institute scientists to present virtual Brain School lectures for March’s Brain Awareness Week , article
Four brain scientists at the Fralin Biomedical Research Institute at VTC will share extraordinary insights and emerging concepts about the brain in one-hour interactive programs.
Date: Mar 09, 2021 -
Article ItemFralin Biomedical Research Institute at VTC scientists identify an elusive link between seizures, cell signaling protein , article
A team of Virginia Tech scientists identified a new link between seizures and deficits in connexin 36, a protein essential to the development of electrical synapses connecting neurons. The discovery, published Jan. 11 in Frontiers of Molecular Neuroscience, found that this interaction may make the brain more prone to having seizures.
Date: Jan 11, 2021 -
Article ItemVirginia Tech scientists link rare medical condition to its cause , article
Using CRISPR genome editing in zebrafish, scientists with the Fralin Biomedical Research Institute at VTC linked an undiagnosed human disease with a rare genetic mutation that causes craniofacial abnormalities.
Date: Apr 30, 2020 -
Article ItemZebrafish study reveals developmental mechanisms of eye movement , article
Zebrafish research is a promising way to understand the neural and genetic causes of eye movement problems in people, according to multi-university research led by Albert Pan of the Fralin Biomedical Research Institute at VTC.
Date: Nov 05, 2019 -
Article ItemVirginia Tech graduate students selected to join competitive Society for Neuroscience program , article
Amnah Eltahir and Alyssa Brunal-Brown, Virginia Tech graduate students at the Fralin Biomedical Research Institute at VTC, were accepted into the Society for Neuroscience’s fellowship and associate programs, respectively.
Date: Jun 11, 2019 -
Article ItemEminent scholar in developmental neuroscience joins Virginia Tech Carilion Research Institute , article
Albert Pan moved his laboratory this summer to join VTCRI. He also serves as the Commonwealth Center for Innovative Technology Eminent Research Scholar in Developmental Neuroscience.
Date: Oct 17, 2017
Recent Media Coverage
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect ItemRoanoke Innovates: A Busy Week on Roanoke's Innovation Corridor , redirect Date: Jan 19, 2021
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item
-
Redirect Item